Your ChemEng research round-up: June 2015
10th July 2015
Since ChemEng365, our new ChemEng blog has become a little quiet - except for a last minute hurrah from Geoff Maitland, see his guest blog 'Five of our Past President's favourite ChemEng365 blogs'.

The ChemEng365 campaign concluded at the end of May when Geoff's term as president ended. But of course, all the amazing chemical engineering research and innovation still goes on. So, it seems only fitting to give you a research round-up on all things chemical and process engineering for the month of June - just in case you missed anything!
Injectable hydrogel could help wounds heal more quickly
A team of chemical engineers from the University of California, Los Angeles (UCLA), US, have a developed a material that creates an instant, superior scaffold that allows new tissue to latch on and grow within the cavities formed between linked spheres of gel.
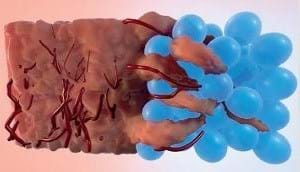
The hydrogel, which is made up of a "scaffold" of microporous annealed particles (MAP gel), fills the wound and promotes swift tissue growth.
"Achieving a biomaterial that promotes rapid regeneration while maintaining structural support has been a holy grail in the field of tissue engineering," explained bioengineering professor Dino Di Carlo.
"Our team has achieved this in an injectable form by combining tailored material chemistry and microfluidic fabrication of uniform spherical building blocks, each about the width of a human hair."
Chemical engineering professor Tatiana Segura also added: "Our technology is beautifully simple, as it utilises any available chemistry to generate tiny gels that can be assembled into a large unit, leaving behind a path for cellular infiltration."
The team recently published their work in the journal Nature Communications.
Chemical engineer wins prize for development of specialised nanotechnology water filter
Dr Askwar Hilonga, a Tanzanian chemical engineer and lecturer at the Nelson Mandela Institution of Science and Technology, has won the first Africa Prize for Engineering Innovation.
The award was made in recognition of the development of a sand-based water filter that uses nanotechnology to clean contaminated drinking water.
Askwar's filter does not require electricity, UV irradiation or chemicals, and releases nothing into the filtered water. It is made up on three components: sand, to remove debris and up to 97 per cent of micro-organisms; the nanofilter, which removes harmful substances such as pesticides and heavy metals; and a hollow-fibre membrane.
Askwar's innovation can be tailored and engineered for a specific body of water and the pollutants it contains.
You can watch Askwar explain his engineering innovation in this YouTube clip:
https://www.youtube.com/watch?v=PNw21Rz37vA
The Africa Prize for Engineering Innovation was launched by the Royal Academy of Engineering (RAEng) in 2014. The prize includes £25,000 which Askwar plans to spend on buying bulk materials to reduce the cost of his filter.
All that's left to say is congratulations to Askwar on his prestigious win!
Nanoscale "designer carbon" could lead to breakthrough in energy storage
Researchers from Stanford University, US, have tested and developed a designer carbon material that has enhanced abilities to catalyse chemical reactions and store electrical charge.

Professor Zhenan Bao, professor of chemical engineering, who led the study said that their designer carbon is both "versatile and controllable". The large porous surface area of this type of material results in increased energy storage capacity in batteries.
The research team developed sheets of carbon just one nanometre thick by fine tuning its properties during manufacture. This result was a material with more than 4,000 square metres of surface area per gram!
Another potential application of designer carbon is for carbon capture technology, Zhenan said: "Our synthetic approach gives us a very versatile, tuneable material, and we are developing a different version of the carbon that’s highly suitable for carbon capture."
Both energy storage and carbon capture are hot topics in today's climate, so it's great to see progress in a research area that looks into these types of advanced materials.
Stretchable conductors could pave way for flexible electronics
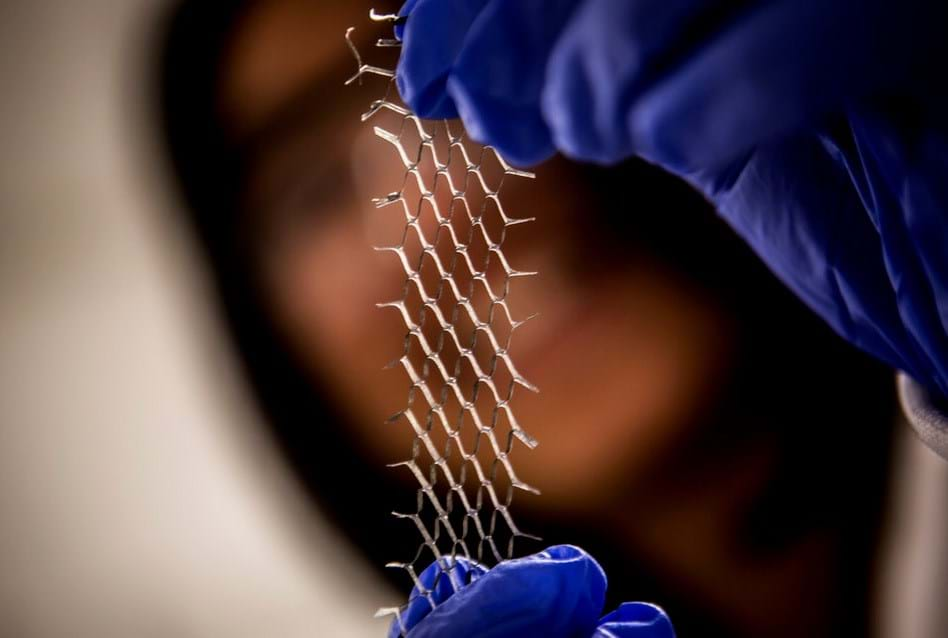
Research engineers have developed a stretchable conductor by using a Japanese cutting and folding technique called kirigami. The team, from the University of Michigan, US, initially created their prototype from tracing paper covered in carbon nanotubes.
Professor Nicholas Kotov, professor of engineering, said: "The kirigami method allows us to design the de-formability of the conductive sheets, whereas before it was very Edisonian process with a lot of misses and not a lot of hits."
When placed in an argon-gas filled glass tube, the paper prototype turned the gas into a glowing plasma. The high voltage across the electrode resulted in electrons in the tube colliding with argon atoms, causing them to emit light.
To understand how design affects the behaviour of stretchable conductors, Sharon Glotzer, professor of chemical engineering, and her research group, performed computer simulations. And they found that stretching their conductors didn't compromise on the material's conductivity.
The multi-disciplinary research team published their findings in the journal Nature Materials. And who knows, their work may pave the way for flexible plasma screens and even flexible smart phones in the future.
More ChemEng research stories from June:
- Student's research helps provide clean water in Palomas, Mexico.
- Chemical vapour deposition enables production of uniform coatings of metals or polymers.
- Research team demonstrate low-cost, environmentally friendly bio-manufacture of quantum dot nano-crystals.
Do you want to share your chemical engineering research? If so, contact the blog elves here and showcase your work.